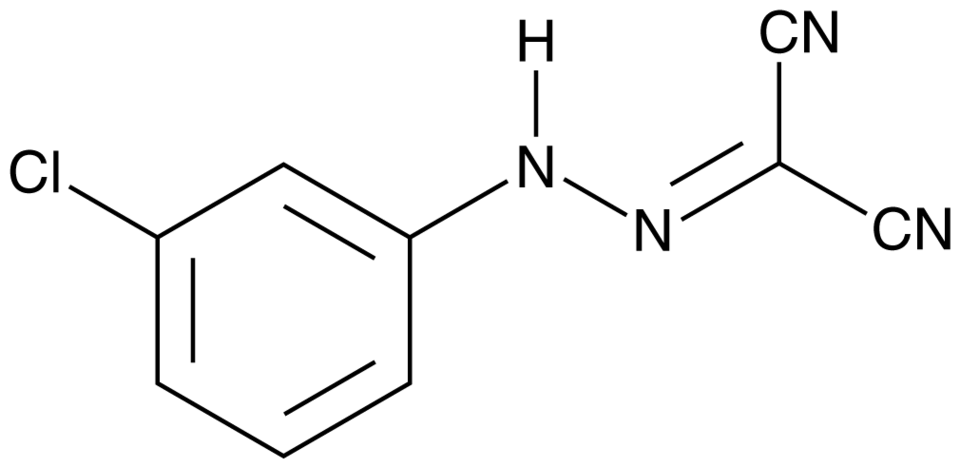
What is cccp cell bio
In addition, ChR2 has been used to manipulate the membrane excitability of cardiomyocytes ( 20– 22), skeletal muscle cells ( 23) and cell lines expressing voltage-gated ion channels ( 24, 25). For example, ChR2 has been expressed in neurons ( 19) to monitor and control their membrane potential, intracellular acidity, Ca 2+ influx, and other cellular processes (see ref ( 14) for review). Due to its high spatiotemporal resolution, ChR2-based optogenetic techniques have evolved as a powerful tool in basic and translational research. When excited by blue light, the all-trans retinal undergoes trans-cis isomerization ( 18), activating non-selective cationic channels and inducing depolarizing inward currents. ChR2 is a seven-transmembrane domain protein that contains the light-isomerizable chromophore all-trans-retinal ( 17). Optogenetics has recently emerged as a technique that utilizes genetically-encoded light-sensitive ion channels, such as channelrhodopsin 2 (ChR2) ( 11– 14) or halorhodopsin (NpHR) ( 15, 16), to precisely and remotely manipulate the activity of cells or animals ( 11, 13). A new approach capable of precisely controlling MMP or ΔΨ m is highly desired for advancing mitochondrial research. A limitation of this approach is that it requires high-energy laser illumination, which may induce uncontrollable oxidative stress and unpredictable outcomes. Several research groups, including us, use laser flashes to trigger local or cell wide mitochondrial depolarization via the photooxidation mechanism ( 7– 10). However, pharmacological approaches often cause unknown side effects and lack the ability to probe spatiotemporal domains. using chemicals to uncouple the mitochondria ( 4, 5) or to induce permeability transition pore (mPTP) opening ( 6). Currently, manipulation of ΔΨ m is mainly via pharmacological intervention, i.e. The detailed mechanism underlying the differential regulatory role of ΔΨ m on cell function and fate remains incompletely understood, partially due to the difficulty of controlling ΔΨ m or mitochondrial membrane permeability (MMP) with spatiotemporal resolution, reversibility, and with cell type specificity in an experimental setting. Interestingly, while profound depolarization of ΔΨ m is detrimental and induces cell injury ( 3), partial mitochondrial dissipation can be cytoprotective ( 4, 5).
The normal functioning of mitochondria largely relies on maintaining the inner membrane potential (ΔΨ m) to preserve the protonmotive force necessary to drive oxidative phosphorylation and redox balance. Not surprisingly, defects of mitochondrial function have been implicated in a variety of human maladies such as aging, cardiovascular disease, cancer, and neurodegeneration ( 2). Mitochondria lie at the crossroad of cellular metabolic and signaling pathways and thus play a pivotal role in regulating many intracellular processes such as energy production, reactive oxygen species (ROS) production, Ca 2+ handling, and cell fate decision ( 1). In summary, we provide evidence that the described mitochondrial-targeted optogenetics may have a broad application for studying the role of mitochondria in regulating cell function and fate decision. Finally, we show that Parkin overexpression exacerbates, instead of ameliorating, mitochondrial depolarization-mediated cell death in HeLa cells. Importantly, we for the first time show that optogenetic-mediated mitochondrial depolarization can be well-controlled to differentially influence the fate of cells expressing mitochondrial ChR2: while sustained moderate light illumination induces substantial apoptotic cell death, transient mild light illumination elicits cytoprotection via mitochondrial preconditioning. We demonstrate successful targeting of the heterologous Channelrhodopsin-2 (ChR2) fusion protein to the IMM and formation of functional cationic channels capable of light-induced selective ΔΨ m depolarization and mitochondrial autophagy. To address this need, we have developed a next-generation optogenetic-based technique for controllable mitochondrial depolarization with light. However, the detailed mechanisms by which ΔΨ m regulates cellular function remain incompletely understood, partially due to difficulty of manipulating ΔΨ m with spatiotemporal resolution, reversibility, or cell type specificity. ΔΨ m) that is essential for ATP synthesis, Ca 2+ homeostasis, redox balance and regulation of other key signaling pathways such as mitophagy and apoptosis.
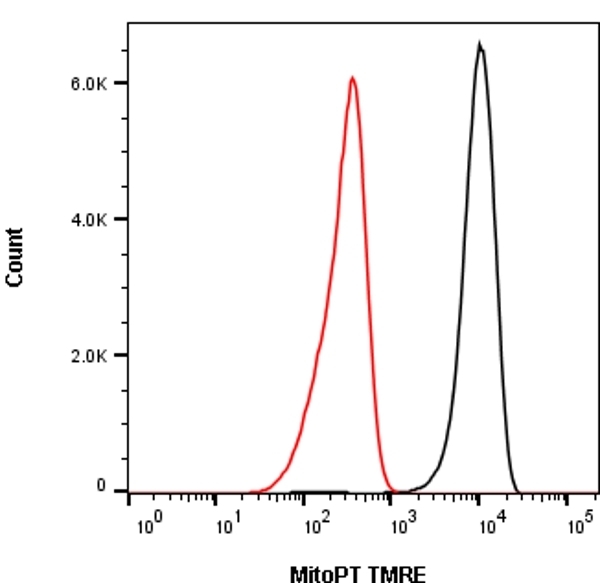
The normal functioning of mitochondria relies on maintaining the inner mitochondrial membrane (IMM) potential ( a.k.a. Mitochondrial dysfunction has been implicated in many pathological conditions and diseases.
